Research outcomes of the Program for the Promotion of Next Generation Research Projects
School of Medicine
Kenji F. Tanaka
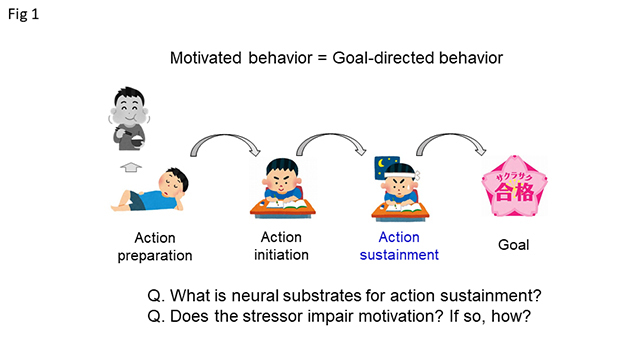
The neural substrates underlying goal-directed action preparation and action initiation have been well characterized, but those underlying action sustainment are poorly understood (Fig. 1). One requirement for sustained goal directed action is suppression of competing negative emotional states. Food seeking, for instance, is potently suppressed by threatening stimuli that evoke fear or anxiety. Successful goal-directed behavior is likely to require gating of the behavioral inhibition associated with negative emotional states.
© Keio University
The hippocampus embedded deep in temporal lobe of the brain is known to monitor the environment, with the dorsal hippocampus (dHP) contributing to spatial and contextual processing and the ventral hippocampus (vHP) regulating emotional responses to internal and external contextual cues. In particular, the vHP modulates threat-related responses, such as anxiety-like behavior and fear. In addition to projecting to the subcortical structures classically associated with these behaviors, the vHP densely projects the ventral striatum, a key orchestrator of goal-directed behavior. The vHP is therefore a potential interface between emotion and goal-directed action.
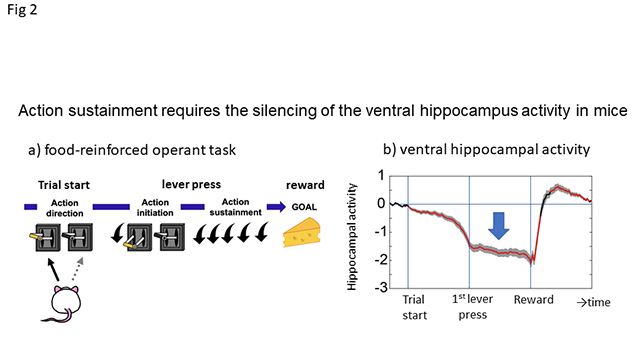
To investigate hippocampal involvement in goal-directed action, we measured the activity of dHP and vHP during food-reinforced operant behavior (Fig. 2a) by training mice to undergo the task of pressing a lever five times to obtain one palatable pellet (fixed ratio (FR)-5 task). We used transgenic mice harboring a Ca2+ indicator in hippocampal CA1 pyramidal neurons (Htr5B-YC mice) and applied fiber photometry to record compound Ca2+ signals from the targeted area in freely moving mice. We employed a Förster resonance energy transfer (FRET)-based ratiometric genetically-encoded Ca2+ indicator, Yellow Cameleon nano50 (YCnano50), which allowed us to reliably monitor both activated and inactivated states as the yellow/cyan fluorescence intensity ratio (YC ratio). Activity in ventral CA1 (vCA1) gradually decreased during the preparatory period (trial start to 1st lever press latency: median 5.5 ± 0.8 sec), was sustained at the lowered level during lever press (1st to 5th lever press latency: median 5.0 ± 0.3 sec) (Fig 2b, arrow), rapidly increased after reward delivery, and then overshot and returned to the baseline. We focused on the inhibition of vHP activity during lever pressing.
© Keio University
We next investigated whether optogenetic artificial inhibition of vCA1 would promote action sustainment. For vCA1 inhibition, we used transgenic mice (Htr5B-ArchT mice), in which only the CA1 pyramidal neurons expressed an inhibitory opsin (ArchT). The vCA1 inhibition reduced completion latency and time-out trials compared with the control in the FR-10 and FR-20 tasks, which require more sustained lever pressing. To further evaluate the effect of vCA1 inhibition on action sustainment, we employed the progressive ratio (PR) task, which progressively increased the number of lever presses required to obtain the reward. As expected, optogenetic vCA1 inhibition from the first to the last lever press increased the maximum number of lever presses achieved in the session, termed the break point. Together these bidirectional manipulations of vCA1 demonstrate that the activity of vCA1 modulates sustainment of goal-directed action.
Impaired motivation is a cardinal symptom of depression. Depressed patients exhibit deficits in effortful reward-based tasks, leading some researchers to conclude that motivational deficits in depressed patients reflect a decreased willingness to expend effort to obtain rewards. Despite this evidence from patient studies, research in animal models for depression has tended to focus on hedonic reward. In two well-validated rodent models for depression, chronic social defeat stress and chronic unpredictable stress, stress-exposed animals display a reduced preference for sweet rewards, which has been interpreted as reflecting blunted hedonic impact. Studies linking stress and motivation have typically focused on striatal regions implicated in hedonic reward processing. There is also evidence that chronic stress can impair goal-directed motivation in operant tasks that require sustained responding to earn rewards. However, the mechanisms of stress effects on goal-directed motivation are unknown.
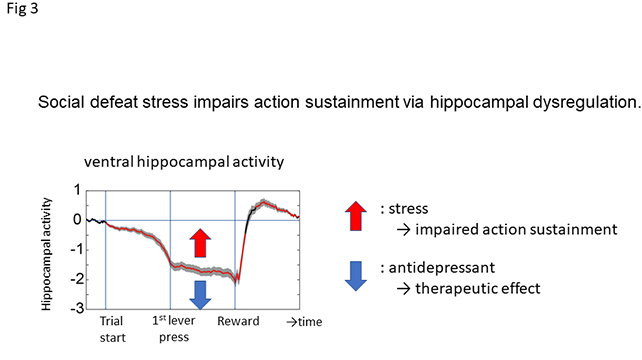
We hypothesized that chronic stress exposure might impair goal-directed motivation by interfering with the suppression of vCA1 activity, and our research demonstrated that social defeat stress impairs goal-directed motivation in mice. However, the population of stress-exposed animals can be stratified into susceptible and resilient populations. Using fiber photometric recordings of bulk neural activity, we have shown that stress effects on goal-directed motivation are associated with perturbations in ventral hippocampal activity (Fig 3, red arrow), and stress-induced motivational impairments can be alleviated with appropriately timed optogenetic manipulation of vHP activity. Finally, we demonstrated that two distinct classes of antidepressants, a slow-acting serotonin selective reuptake inhibitor (SSRI) and rapid/prolonged-acting ketamine, rescue stress-induced impairments in motivated behavior and hippocampal function (Fig 3, blue arrow).
© Keio University
In summary, we identified that the vHP was a key domain of action sustainment of goal-directed behavior. The silencing of vHP was necessary for successful goal-directed action sustainment to attain the reward in healthy conditions. During repeated social defeat stress, stress-susceptible mice showed that vHP silencing was gradually perturbed. Dysregulation of vHP activity can be a circuity biomarker of impaired motivation and its normalization should be the therapeutic target.
Reference
- Yoshida K, Drew MR, Mimura M, Tanaka KF. Serotonin-mediated inhibition of ventral hippocampus is required for sustained goal-directed behavior. Nature. Neuroscience, 22(5), 770-777, 2019
- Yoshida K, Drew MR, Kono A, Mimura M, Takata N, Tanaka KF. Chronic social defeat stress impairs goal-directed behavior through dysregulation of ventral hippocampal activity in male mice. Neuropsychopharmacology. 46(9):1606-1616, 2021
Keio University Program for the Promotion of Next Generation Research Projects
The Keio University Program for the Promotion of Next Generation Research Projects subsidizes research costs with the aim of finding solutions to challenges and of promoting global academic research in order to allow Keio University faculty members to establish a presence as core researchers.