Cyclic organic molecules play important roles in living organisms and serve as inspiration for the design of functional synthetic compounds. Now, two such ring-like molecules have been successfully synthesized, with possible applications including sensors and catalysts.

©︎ Damerfie ― stock.adobe.com/jp
Cyclic compounds are molecules in which some of the atoms are connected to form a closed ring. Porphyrins are an important naturally occurring class of such cyclic compounds, and hemoglobin, carrying oxygen around in the human body, and chlorophylls, enabling photosynthesis in plants, are well-known organic molecules based on the porphyrin structure. Given their key roles in living organisms, porphyrins have inspired the design of synthetic compounds for various applications. Recently, Naoya Kumagai from Keio University and colleagues have succeeded in synthesizing two new members of the porphyrin family, with promising physicochemical properties such as fluorescence and catalytic activity.
The first molecule synthesized by the researchers is called TEtraQuinoline (TEQ). The molecule's name refers to quinoline, a small organic molecule consisting of carbon, nitrogen and hydrogen atoms; TEQ consists of 4 connected quinoline units, in which the central ring contains 4 nitrogen atoms as well as 12 carbon atoms. The molecule has a rigid, saddle-like, three-dimensional structure. Due to this rigid structure, the compound could not be synthesized by 'simply' connecting the quinoline units, but rather by first constructing a cyclic precursor, of which particular parts were then subsequently chemically modified.
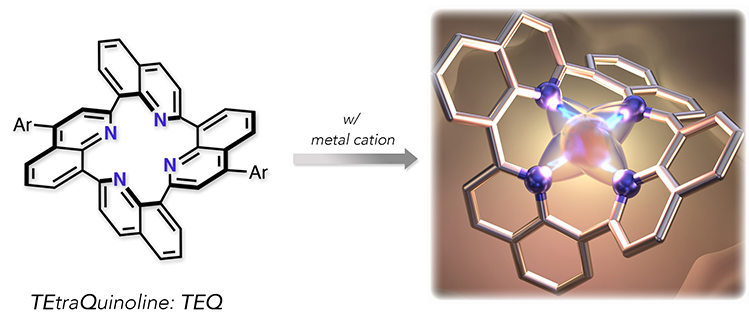
[Fig. 1]
Structure and metal complexation of TEtraQuinolines (TEQs)
TEQ turned out to have remarkable physicochemical properties. By itself, the molecule displayed only low fluorescence (the absorption of light, followed by the emission of light with a lower frequency). But in an acidic environment, fluorescence became high, a property that may be used for developing acidity level (pH) probes. Likewise, when TEQ was brought in contact with zinc ions, its fluorescence increased significantly, a discovery that may lead to potential zinc-sensing applications. TEQ was found to form complexes not only with zinc, but with a range of other metals too. The TEQ-iron complex was found to exhibit high catalytic activity at very low concentration, which opens the door for its use as a catalyst in important chemical processes such as carbon dioxide reduction and photochemical water splitting.
The second molecule synthesized by Kumagai and colleagues is Oxa-TriQuinoline (o-TQ) and, as the name suggests, it is also based on the quinoline unit. It is a modification of TriQuinoline ― which consists of 3 connected quinoline units ― featuring a central ring containing 3 oxygen, 3 nitrogen and 9 carbon atoms. o-TQ's structure is semi-rigid and has a bowl-like shape.
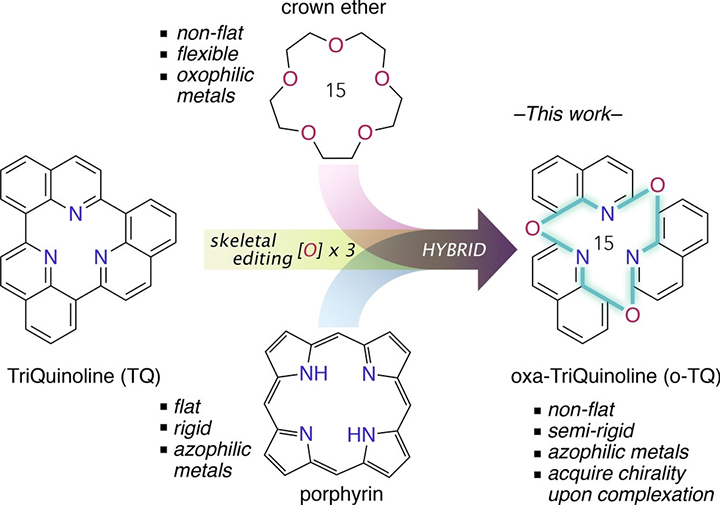
[Fig. 2 from the paper2]
from the paper2]
Inspiration for the design of the 15-membered macrocycle oxa-triquinoline (o-TQ)
(https://doi.org/10.1002/anie.202307896) is licensed under CC BY NC 4.0 DEED
The scientists investigated several properties of the newly synthesized o-TQ. They found that o-TQ forms a complex with copper and that this complex exhibits 'solid-state photoluminescence'― particularly, the emission of orange light. The researchers also studied the catalytic potential of o-TQ, and discovered that the o-TQ-copper complex is catalytically active in certain organic reactions.
The successful synthesis of TEQ and o-TQ reported in this research offers new approaches for establishing innovative methods for the creation of next-generation functional molecules and related applications.
Quoting Kumagai and colleagues: "... the rigid and robust tetradentate ligand TEQ represents a new platform for catalyst design and molecular engineering." They go on to say that "the rich chemistry elicited from the newly designed cyclic tridentate ligand [o-TQ] ... can be expected to inspire further exploration in molecular design to pursue hitherto unknown molecular functions."
Published online 13 March 2024
About the researcher

Naoya Kumagai ― Professor
Faculty of Pharmacy
Naoya Kumagai earned a Bachelor, Master, and PhD in Pharmaceutical Sciences from the University of Tokyo in 2000, 2002, 2005, respectively. He is currently a Professor at the Graduate School of Pharmaceutical Sciences, Keio University.
His research interests are in organic chemistry, materials chemistry, and interdisciplinary research between chemistry and biology.
Links
Reference
- 1. Xu, W., Nagata, Y. & Kumagai, N. TEtraQuinolines: A Missing Link in the Family of Porphyrinoid Macrocycles. J. Am. Chem. Soc 145, 2609-2618 (2023). | article
- 1. 2. Kobayashi, T. & Kumagai, N. Oxa-TriQuinoline: A New Entry to Aza-Oxa-Crown Architectures. Angew. Chem. Int. 62, e202307896 (2023). | article